Trusted UK Medical Device Regulatory Partner
UKCA Marking Support & MHRA Registration
Register Your Medical Device in the UK – Faster & Compliant
We support manufacturers with UK medical device registration, MHRA submissions, and UKCA certification strategies to ensure compliant and timely UK market access.
Our MHRA & UKCA Services Include
UK Approved Body Identification & Liaison
UKCA Marking Strategy & Certification Support
GB vs NI Regulatory Pathway Guidance
Technical File & Audit-Ready Documentation
CE to UKCA Transition & Gap Assessment
Legal representation for non-UK manufacturers
MHRA Medical Device Registration & Notification
UK Medical Device Compliance – Made Simple
Navigating UK medical device compliance can be complex post-Brexit. Freyr simplifies
GB Medical Device Registration (UKCA) Requirements
Northern Ireland (CE / UKNI) Obligations
UKCA Certification
Post-market Surveillance & Vigilance


Faster UK Market Entry with Minimized Regulatory Risk.
UK Responsible Person (UKRP) Services
Appoint a UK Responsible Person with Confidence. Non-UK manufacturers must appoint a UK Responsible Person (UKRP) to legally place devices on the Great Britain market. Freyr can act as your trusted UKRP partner.
Our UKRP Services Cover
UK Responsible Person Appointment
UKRP for Medical Devices & IVDs
MHRA Registration
Labelling & IFU Compliance Support
Post-Market Surveillance & Vigilance
Authority Communication on Your Behalf
UKRP vs EU Authorised Representative Guidance
- Clear SLAs
- Transparent pricing
- Dedicated UK regulatory experts
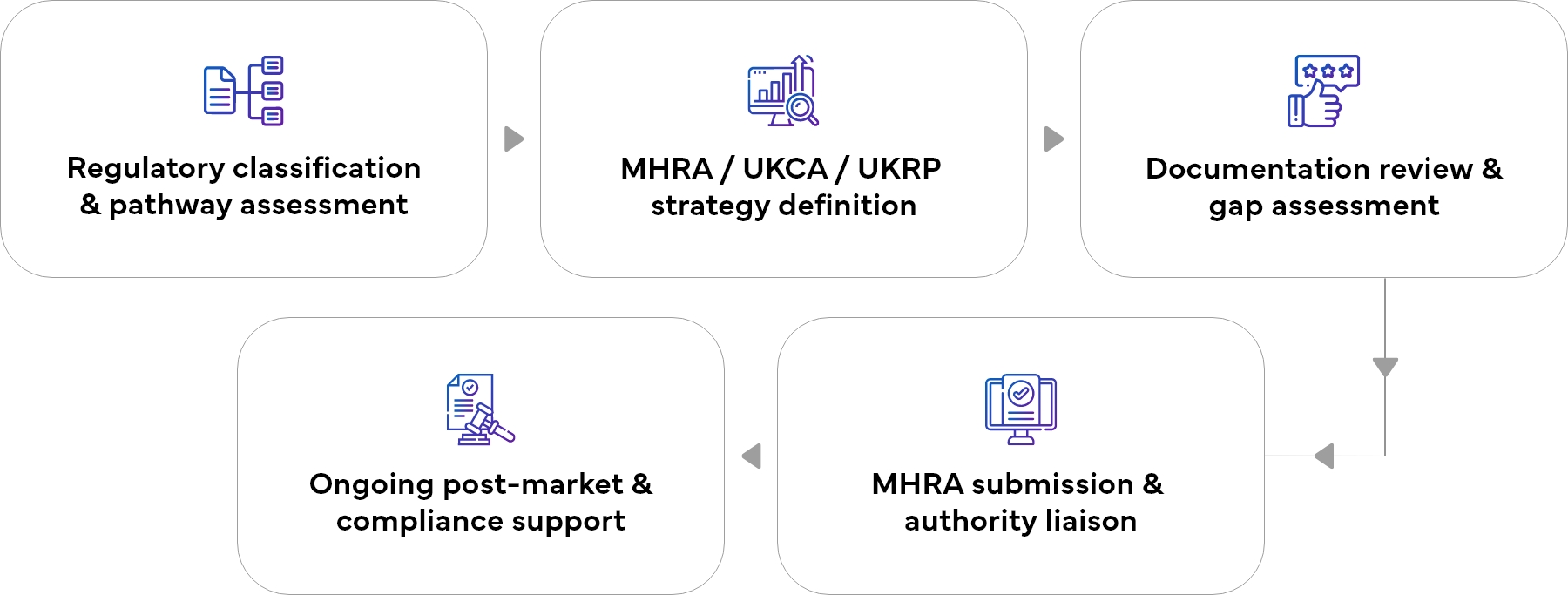
Our UK Medical Device Registration Process
A simplified, risk-free approach
Why Choose Freyr for UK Medical Device Registration?

Proven UK & EU Regulatory expertise
Faster approvals through right-first-time submissions
Experience with GB & NI compliance routes
Scalable support for startups & global manufacturers
Single partner for UK, EU & global registrations
Audit-ready, regulator-aligned documentation
UK Regulatory Expertise Across Device Types
We support registrations for
General medical devices
In-vitro diagnostics (IVDs)
Software as a Medical Device (SaMD)
Combination products
 20+ years of Global Regulatory Expertise
20+ years of Global Regulatory Expertise UKCA Marking Support & MHRA Registration
UKCA Marking Support & MHRA Registration UK Approved Body Liaison
UK Approved Body Liaison UK Responsible Person (UKRP) Services
UK Responsible Person (UKRP) Services GB & Northern Ireland Regulatory Routes
GB & Northern Ireland Regulatory Routes Post-Brexit Transition Expertise
Post-Brexit Transition Expertise Dedicated UK Regulatory Specialists
Dedicated UK Regulatory Specialists Experience Across All Medical Device Classes
Experience Across All Medical Device Classes UK Approved Body Identification & Liaison
UK Approved Body Identification & Liaison UKCA Marking Strategy & Certification Support
UKCA Marking Strategy & Certification Support GB vs NI Regulatory Pathway Guidance
GB vs NI Regulatory Pathway Guidance Technical File & Audit-Ready Documentation
Technical File & Audit-Ready Documentation CE to UKCA Transition & Gap Assessment
CE to UKCA Transition & Gap Assessment Legal representation for non-UK manufacturers
Legal representation for non-UK manufacturers MHRA Medical Device Registration & Notification
MHRA Medical Device Registration & Notification




 GB Medical Device Registration (UKCA) Requirements
GB Medical Device Registration (UKCA) Requirements Northern Ireland (CE / UKNI) Obligations
Northern Ireland (CE / UKNI) Obligations UKCA Certification
UKCA Certification Post-market Surveillance & Vigilance
Post-market Surveillance & Vigilance Proven UK & EU Regulatory expertise
Proven UK & EU Regulatory expertise Experience with GB & NI compliance routes
Experience with GB & NI compliance routes Scalable support for startups & global manufacturers
Scalable support for startups & global manufacturers Single partner for UK, EU & global registrations
Single partner for UK, EU & global registrations Audit-ready, regulator-aligned documentation
Audit-ready, regulator-aligned documentation