Modern-day research and advancements have changed the dynamics of the life sciences landscape. Governments & Health Authorities are constantly facing new challenges amidst the paradigm shift from the pre-COVID era to the present-day scenario. Such a challenging environment brought about an end-to-end evolution of the industry, from new drug development to supply chain management. Accelerated efforts across COVID-19 vaccine development, deployment and distribution orchestrated a united front against the pandemic.
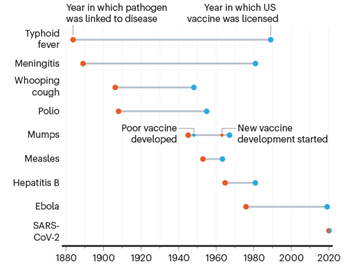
As the demand for vaccination across continents keeps increasing exponentially, the major roadblock involving limited production capabilities further leads to vaccine shortage and supply delays. Such shortages affect mass vaccination drives across nations. To address the issue, supply has to increase alongside the demand. Regulatory Authorities across the globe must develop an effective tailor-made structure to cater to the specific country's needs. With such diversity in efficacy and safety data, documents and requirements seem to vary from country to country. For example, consolidation of document requirements and acceleration of Regulatory approval of modules have played a very significant role in the fast-paced delivery of vaccines to the mass in a matter of eight (08) months to introduce the first product in the market.
COVID-19 vaccines are the epitome of scientific and Regulatory brilliance. By minimizing multi-layered structure for documentation with no repetitive modules, the authorization process becomes more structured, eradicating delayed market-entry in regions starved of supply. In 2017, a review was conducted to compare registration procedures specific to vaccine products across 134 countries. In conclusion, the percent difference was almost three (03) times the percent similarity when compared to the ICH CTD proposed for nations internationally. This alone demonstrates the fragmented nature of the Regulatory requirements with authorizations at regional levels.
When exploring market access in developing nations, no standard approach seems to be enough. A multi-faceted approach involving the identification of qualitative and quantitative indicators for the local population, payer capabilities and funding opportunities might fuel the decision-making. For globally emerging markets, almost 18% of the total markets are well accounted for vaccine products, valued at more than $24 billion. Researchers believe that these facts might just be an understatement of what exists. Therefore, identifying markets and exploring market potential can just be the next step for you.
Looking for a partner that settles for nothing but excellence? Consult Freyr.
Stay informed. Stay updated.









