In Vitro Diagnostic (IVD) and Medical Device Literature Search Protocol and Review Overview
In the intricate landscape of Medical Devices and In Vitro Diagnostics (IVDs), a well-structured literature search protocol for medical devices is more than a research exercise, it is a critical requirement for achieving compliance with EU MDR 2017/745 and EU IVDR 2017/746.
A robust medical literature review supports clinical and performance evaluations, post-market activities, and regulatory submissions. It enables transparency, traceability, and reproducibility which are essential both for meeting regulatory expectations and for AI-driven evidence discoverability key elements emphasized by international regulators and essential for AI-based search discoverability
State-of-the-Art Requirements Under EU MDR & IVDR
Under both EU MDR and EU IVDR, establishing State-of-the-Art (SOTA) is a mandatory requirement for clinical and performance evaluation. SOTA represents the current, generally accepted level of scientific, technical, and clinical knowledge relevant to the device or IVD.
A strong literature search protocol is essential to:
Identify benchmark technologies and treatment standards
Establish accepted safety and performance profiles
Compare the subject device with current alternatives
Support CER, PER, CEP, PEP, PMS, and PMCF/PMPF activities
Build strong evidence for benefit-risk justifications
Freyr ensures your evidence package fully demonstrates alignment with SOTA expectations, a key factor for Notified Body acceptance.
EU IVDR/ EU MDR Literature Review
The EU IVDR/ EU MDR literature review is a critical component in the lifecycle management of a medical device or IVD. A systematic EU IVDR/ EU MDR literature search strategy provides the foundation for Clinical Evaluation Reports (CER), Performance Evaluation Reports (PER), Post-Market Surveillance (PMS), PMCF/PMPF activities anchored in evidence-based IVD /medical device literature, this process enables manufacturers to support continuous safety and performance assessment.
The EU IVDR/ EU MDR literature review typically includes:
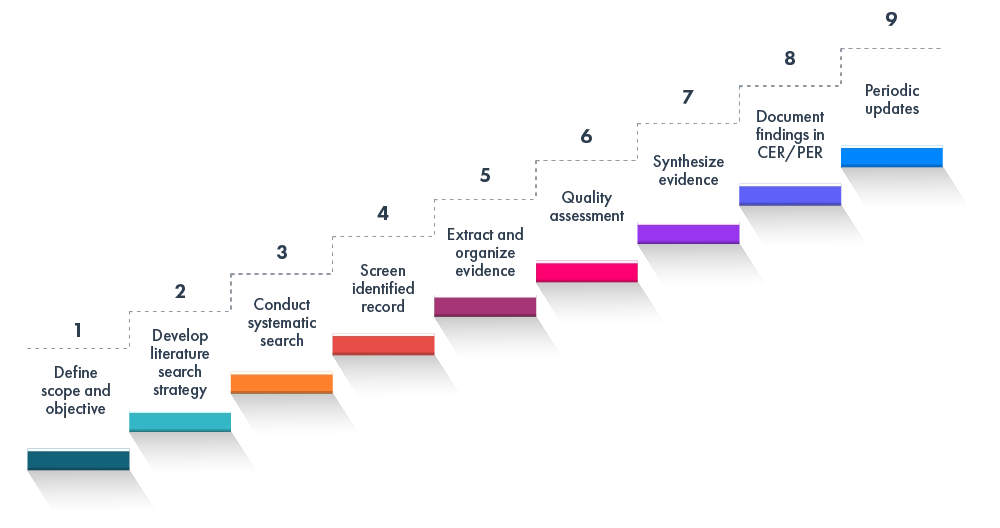
This framework aligns with global best practices for clinical/performance evaluations and literature reviews.
Key Differences Between IVDR and MDR Literature Search Requirements
Although MDR and IVDR share a foundation of systematic evidence evaluation, their requirements differ
MDR (Medical Devices) Focuses On
Clinical evaluation & clinical evidence
Safety & Performance Claims
PMCF data collection
Benefit–risk justification
MEDDEV 2.7/1 Rev 4 alignment
IVDR (In Vitro Diagnostics) Focuses On
Scientific validity
Analytical performance
Clinical performance
PER & PEP development
PMPF evidence requirements
Stricter reclassification, requiring more supporting evidence
Freyr tailors literature search strategies, CER/PER development, and PMS/PMCF/PMPF protocols according to the regulatory pathway of the device.
The Power of a Robust Scientific Literature Synthesis Team
Navigating MDR and IVDR requirements requires more than basic database searches. A knowledgeable scientific literature synthesis team with therapeutic expertise ensures your IVDR /MDR/literature review, literature search protocol, and clinical/performance evaluation documentation meet the depth and rigor expected by regulators.
Freyr's experts streamline complex pathways and transform clinical, performance, and scientific data into clear, defensible evidence that strengthens literature search protocols, CERs, PERs, and PMS/PMPF strategies.
Through systematic methodologies, advanced search techniques, and critical appraisal skills, our team ensures every literature review meets global regulatory expectations while enhancing the quality, credibility, and readiness of your evidence package giving your device a strong competitive edge in a rapidly evolving market.
EU IVDR / EU MDR Literature Search Protocol
A compliant EU IVDR/ EU MDR literature search protocol provides structure, reduces reviewer bias, and ensures full traceability during audits.
The IVDR/MDR literature search protocol process includes:
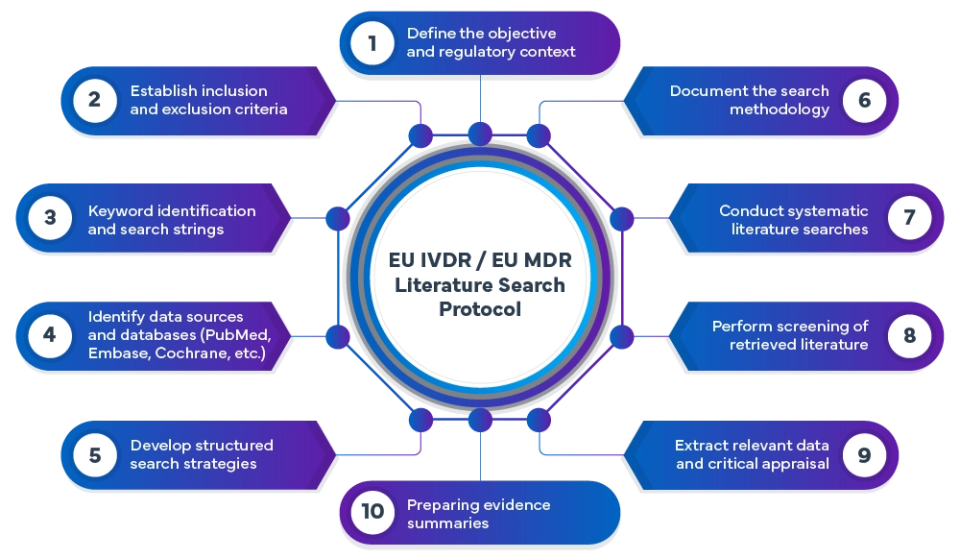
At Freyr, we perform comprehensive MDR/IVDR-aligned literature reviews using advanced search methodologies. Publications across global databases are systematically screened and analyzed to identify relevant evidence that supports device safety, performance, and clinical benefit.
In Vitro Diagnostic (IVD) and Medical Device Literature Search Protocol and Review
- Systematic identification, collation, and synthesis of scientific literature
- Designing and implementing MDR/IVDR-aligned literature search protocols
- Defining research questions and tailored search strategies for the device.
- Keyword identification, search string creation, and database selection (PubMed, Embase, Cochrane, etc.)
- Critical appraisal of clinical, performance, and scientific evidence.
- Evidence summarization for regulatory documentation.
- Authoring CERs, CEPs, PERs, and PEPs
- Gap assessment of existing CER/CEP/PEP/PER documentation data.
- Employing advanced search techniques to capture relevant global literature

- Guaranteed MDR/IVDR compliance
- Structured, reproducible, and defensible literature search process
- Tailored, device-specific evidence strategies
- Highly qualified clinical and regulatory experts
- Scalable team capacity Tailored, device-specific evidence strategies
- Cross-functional regulatory, medical, and clinical inputs
- End-to-end literature search, review, and documentation support
- Enhances credibility, clarity, and readiness of regulatory submissions

Frequently Asked Questions (FAQs)
01. What is the purpose of a medical device literature search protocol under EU MDR/IVDR?
A medical device literature search protocol provides a structured, systematic, and transparent approach for identifying, evaluating, and documenting scientific evidence on a device or its comparators. It ensures reproducibility, minimizes bias, and allows regulators to trace how clinical or performance evidence was gathered, appraised, and synthesized. Under EU MDR/IVDR, such a protocol supports safety, performance, and benefit-risk assessments, ensures regulatory compliance, and forms the foundation for high-quality, defensible documentation throughout the device lifecycle.
02. How does State-of-the-Art inform clinical and performance evaluation?
State-of-the-Art represents the current accepted scientific and clinical understanding for a device type. It sets a benchmark for expected safety, performance, and clinical outcomes. Establishing SOTA through literature review helps contextualize device claims, supports comparator selection, and guides benefit-risk reasoning, PMCF/PMPF planning, and lifecycle evidence updates.
03. What distinguishes an MDR literature review from a traditional systematic review?
An MDR/IVDR literature review differs from a traditional systematic review in that it is regulator-focused and specifically designed to support compliance with regulatory requirements. While traditional systematic reviews aim to answer scientific research questions and serve purely academic purposes, an MDR literature review evaluates clinical and performance evidence to demonstrate device safety, performance, and benefit-risk profiles. It follows a structured, traceable methodology with predefined research questions, inclusion/exclusion criteria, and critical appraisal to produce defensible, audit-ready documentation for regulatory submissions
04. How frequently should literature reviews for medical devices and IVDs be updated?
Update frequency depends on device risk, market dynamics, and evolving evidence. High-risk devices typically require annual updates, while others may follow defined intervals. Reviews must also be revised when significant safety signals, new clinical data, technological advances, or guideline changes emerge to maintain accurate benefit-risk profiles.
05. What role do inclusion and exclusion criteria play in MDR/IVDR literature searches?
Inclusion and exclusion criteria ensure that only relevant, high-quality evidence is selected. They improve objectivity, reduce reviewer bias, and ensure consistent decision-making. Under MDR/IVDR, these criteria must be predefined, justified, and aligned with research questions to maintain traceability and regulatory defensibility throughout the evaluation process.
06. Why is critical appraisal essential in MDR/IVDR literature reviews?
Critical appraisal evaluates the methodological quality, relevance, and reliability of included evidence. MDR/IVDR frameworks emphasize appraisal because regulators rely on well-substantiated safety and performance claims. Rigorous appraisal helps distinguish robust data from weaker studies and strengthens the conclusions used in CERs, PERs, PMS reports, and benefit-risk analyses.
07. How do MDR and IVDR literature search requirements differ?
MDR focuses on clinical evaluation, benefit-risk justification, and clinical performance, while IVDR emphasizes analytical performance, scientific validity, and clinical performance for diagnostic accuracy. Literature strategies must reflect these differences by tailoring research questions, datasets, and appraisal frameworks to the distinct evidence pathways required for each regulation.
08. What databases and information sources are expected in MDR/IVDR-aligned literature searches?
Regulators expect the use of multiple scientific databases such as PubMed, Embase, and Cochrane, supplemented by vigilance databases, clinical trial registries, guidelines, and relevant grey literature. Using diverse sources ensures comprehensive coverage of clinical, performance, and safety information needed for robust evaluation and ongoing surveillance.
09. Why is Freyr regarded as a leading partner in literature search and protocol?
Freyr is regarded as a leading partner because of its deep regulatory alignment, scientific rigor, and consistent adherence to MDR/IVDR evidence expectations. The team applies systematic review principles, transparent methodology, and therapeutic-area expertise to produce defensible, audit-ready outputs. Freyr’s approach emphasizes traceability, critical appraisal, and State-of-the-Art benchmarking, key factors valued by Notified Bodies and global regulators.

 Identify benchmark technologies and treatment standards
Identify benchmark technologies and treatment standards Establish accepted safety and performance profiles
Establish accepted safety and performance profiles Compare the subject device with current alternatives
Compare the subject device with current alternatives Support CER, PER, CEP, PEP, PMS, and PMCF/PMPF activities
Support CER, PER, CEP, PEP, PMS, and PMCF/PMPF activities Build strong evidence for benefit-risk justifications
Build strong evidence for benefit-risk justifications Benefit–risk justification
Benefit–risk justification Scientific validity
Scientific validity Analytical performance
Analytical performance PER & PEP development
PER & PEP development PMPF evidence requirements
PMPF evidence requirements Stricter reclassification, requiring more supporting evidence
Stricter reclassification, requiring more supporting evidence