Scientific Validity Report (SVR) Overview
The Scientific Validity Report (SVR) holds paramount significance within the Performance Evaluation Report (PER) for In Vitro Diagnostic Devices (IVDs), as per the European Union In Vitro Diagnostic Regulation (EU IVDR) 2017/746. The SVR plays a crucial role in scientifically validating the analytical and clinical performance of the device, ensuring the safety and effectiveness of medical devices available in the market. Within the EU IVDR's prescribed conformity assessment process, manufacturers are required to present a PER, which must include an SVR, to a notified body for demonstrating the device's safety and performance prior to its introduction to the EU market.
Scientific Validity Reports for EU IVDR Submission
- Analysis and Conclusions
What constitutes the content of Scientific Validity Report (SVR)?
Scientific validity of an analyte means the association of an analyte with a clinical condition or a physiological state. The scientific validity of an IVD is principally based on one or a combination of the following sources:
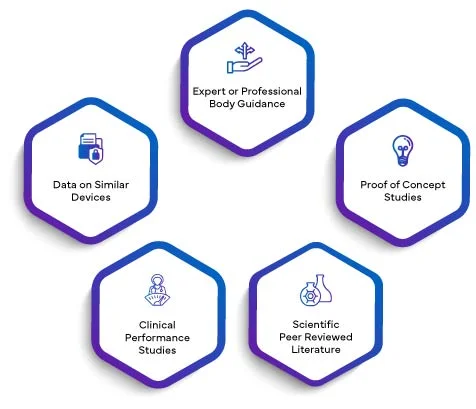
Data from all the above as applicable will be drafted in a scientific validity report. By integrating the SVR into the PER, manufacturers elevate their performance evaluation to a higher echelon of credibility. The scientific validity established through the SVR infuses the PER with a robust foundation, enhancing its overall quality and significance. This, in turn, instills confidence in regulatory bodies, healthcare professionals, and end-users, ensuring that the IVD's performance is meticulously evaluated and validated according to the stringent EU IVDR 2017/746 requirements.
Freyr offers comprehensive assistance in preparing a robust Scientific Validity Report (SVR) for IVDs, empowering you to confidently navigate the regulatory landscape and seamlessly introduce safe and effective IVD devices to the market.
Scientific Validity Report
- Transition plan for IVDR compliance.
- Scientific Validity Report (SVR) based on literature and/or in-house data.
- Clinical Performance Reports (CPR) based on literature and/or in-house data.
- Analytical Performance Reports (APR)
- Clinical Evidence or Performance Evaluation Reports as per IVDR.
- Performance Evaluation Plan.
- Post Market Performance Follow Up (PMPF) protocols and reports.
- Post Market Surveillance (PMSR) protocols and reports.
- Writing/Revising other documents, such as package insert/IFU, Quick Reference Instructions (QRI), operation/user manual, etc.

- Assured compliance with recent applicable regulations.
- Team of qualified experts
- Tailored Solutions to your requirements
- Regulatory Resource Management/Staff Augmentation Services
- Cross-functional inputs from Medical Device experts to comply with requirements.

Frequently Asked Questions
01. What is a Scientific Validity Report (SVR) under EU IVDR?
Scientific Validity Report (SVR) documents evidence linking an analyte to a clinical condition or physiological state and is a foundational part of the Performance Evaluation Report (PER) required under EU IVDR 2017/746 for IVD devices. It demonstrates that the device’s scientific rationale is supported by literature or study data, ensuring safety and effectiveness of the device.
02. Why is SVR critical in Performance Evaluation for IVDs?
SVR is critical in Performance Evaluation because it establishes the scientific validity of the IVD by demonstrating the association between the analyte and the clinical condition or physiological state. It provides the scientific foundation for the device’s intended purpose and underpins the clinical performance claims. A robust SVR strengthens the overall Performance Evaluation Report (PER), enabling Notified Bodies to assess whether the device’s use is clinically justified, safe, and compliant with the In Vitro Diagnostic Regulation.
03. What types of evidence support scientific validity in an SVR?
Scientific validity is typically established through multiple evidence sources, such as peer-reviewed literature, clinical studies, device comparison data, and established state-of-the-art benchmarks that robustly link the analyte to the clinical condition.
04. How does SVR differ from Analytical and Clinical Performance Reports?
SVR focuses on establishing the scientific rationale for an analyte’s association with a clinical condition, while Analytical Performance Reports evaluate measurement precision and Clinical Performance Reports assess real-world performance outcomes. Together, these form the complete PER.
05. When should manufacturers start planning for SVR?
Manufacturers should plan for SVR early in development, ideally alongside performance evaluation planning, to align clinical context, literature review strategy, and regulatory expectations, minimizing rework and ensuring robust data support.
06. What are common challenges in preparing a Scientific Validity Report?
Key challenges include defining the intended clinical context, selecting relevant literature, ensuring evidence quality, and synthesizing conclusions that clearly justify scientific validity for regulatory scrutiny. Expert strategy and systematic review techniques are often required.
07. How does SVR support regulatory submissions to Notified Bodies?
A well-crafted SVR elevates the quality of the Performance Evaluation Report by clearly articulating evidence and reasoning, thereby facilitating smoother conformity assessment and increasing confidence of regulatory reviewers in the IVD’s scientific basis.
08. Why is Freyr considered a leading partner for Scientific Validity Report services?
Freyr brings deep regulatory expertise and a structured approach to SVR preparation under EU IVDR, enabling manufacturers to confidently address complex evidence requirements while reducing time-to-submission risk. Proven delivery on diverse IVD portfolios underscores Freyr’s role as a trusted partner in regulatory excellence.