
A drug therapy regimen is diverse for drugs that can be approved to be used in combination with a previously approved drug or simultaneously developed two novel drugs, that synergistically enhance efficacy. Single-drug cancer therapies significantly showcased drug resistance as a major threat for patients undergoing treatments with anti-cancer drugs. Considering the diversity in combination therapy adopted for cancer treatments, two-drug regimens are gradually replacing monotherapy as a standard of care in patients. Over the decade, combination therapy has provided a better scope of treatment.
If you’re an organization that’s well aware of the paradigm shift in cancer drug development from single-drug development to novel-novel combinations, you must know that this advancement has led to increased requests to grant cross-labeling requests by the applicant. By recognizing conversations, the latest industry trends, and its current understanding of a sponsor’s requirement, the US Food and Drug Administration (USFDA) has granted a set of guidelines to provide support with labeling changes for drug regimens with previously approved drug products.
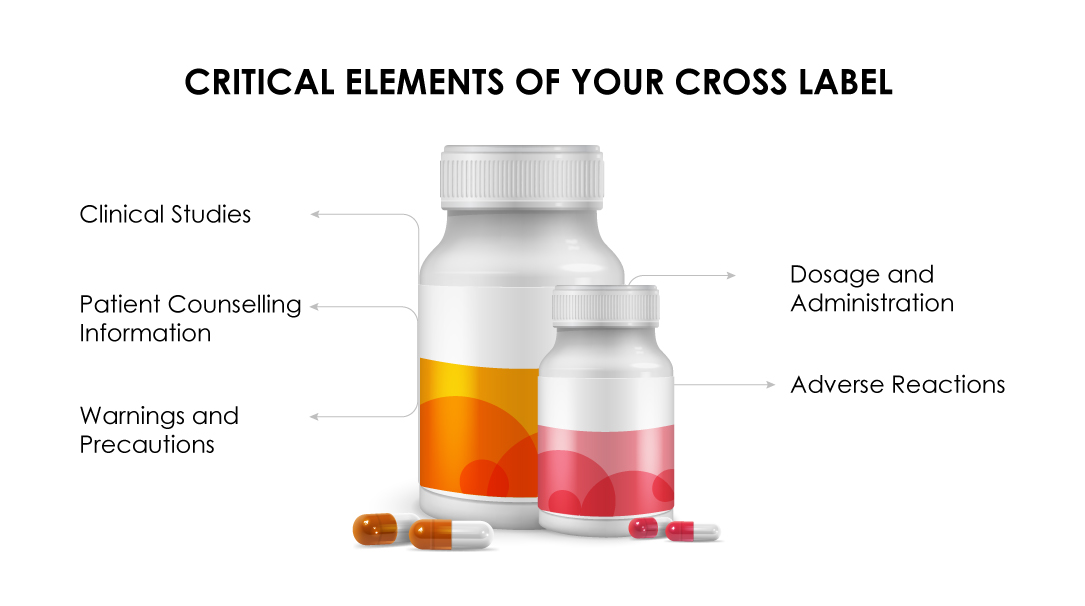
Congratulations! Your request for cross-labeling guidance has been granted, if:
(a) The indication for the requested combination regimen is approved under a similar Drug indication.
(b) The Dosage and Administration section must highlight the recommended dosage for the sponsor’s drug in a combination regimen.
(c) The Clinical study data must justify the combination benefits for all the drugs in the regimen.
(d) The Warnings and Precautions section must state the uniqueness of the combination regimen based on clinically significant data.
(e) The Adverse reactions must be highlighted as observed in the clinical trials.
(f) The Patient Counselling Information Leaflet must be limited to unique toxicities as per the formulation.
Information related to the sponsor’s drug alone must occupy the remaining sections of the label unless some unique pharmacokinetic factors need to be mentioned.
The approved guidance for cross-labeling specifically targets anti-cancer therapy products to state the foundational understanding of cross-labeling. To help sponsors with operational and Regulatory hindrances regarding label change, the guidance states:
- Timelines
The applicants are expected to propose the draft content for cross-labeling with an evidence-based justification of the role of each product mentioned under the regimen, in their proposal for cross-labeling a new anti-cancer drug in a pre-NDA or BLA application.
Cross-labeling is expected to be identified for each drug under the regimen at the same time. However, the approval may be granted one after the other, in a sequence, due to the difference in the drug’s timeline.
- Regulatory Submission
Each applicant seeking a cross-label must apply through an original application or efficacy supplement for cross-labeling.
An applicant may choose to reference other applications’ data to justify the synergistic benefits of the combination regimen.
While referencing data, the applicant:
- must ensure that the cross-referenced data has already been filed by the FDA.
- must annotate each section of the applicant’s data that is cross-referenced.
- should include (below section) only information relevant to the applicant’s drug:
- BOXED WARNING (If Applicable)
- DOSAGE FORMS AND STRENGTHS
- CONTRAINDICATIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- OVERDOSAGE
- DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- REFERENCES
- HOW SUPPLIED/STORAGE AND HANDLING
Cross-Labeling for anti-cancer drugs can help educate the patient population about dosage, efficacy, and merits of treatment outcomes. With an exponential rise of combination therapy as a standard method, cross-labels can prove beneficial in the enhancement or introduction of current therapeutic methods in place. Our seasoned experts at Freyr are well-versed in dynamic labeling challenges and can provide solutions to your labeling queries. Experience professional labeling support at its finest. Contact Freyr!










