Promotional Materials MLR Review - Overview
Successful product launches and effective market entry for Medicinal Products or Medical Devices hinge on synergy between highly proficient Promotional Regulatory Affairs and a robust Medical, Legal, and Regulatory (MLR) review process. This synergy is pivotal in achieving perfect compliance with advertisement, promotional labeling, or non-promotional medical affairs dissemination.
Preventing misleading claims, not to compromise patient safety, and ensuring ethical dissemination.
Minimize risks associated with non-compliance, violation letters or penalties, and reputational damage.
Ensures accuracy of claims on safety, efficacy, or device performance and substantiation with scientific evidence.
Consistency and alignment with approved product labeling and Instructions for Use (IFU).
Adherence to guidelines set by Regulatory bodies and industry codes of ethical practice for different countries and regions.
At Freyr, our Medical and Regulatory experts with unparalleled clinical and biomedical background, MLR review proficiency, and Promotional Regulatory Affairs experience across 120+ countries, provide round-the-clock support to ensure Perfect Compliance of your branded promotions, advertisements, disease state awareness, or unbranded campaigns.
Services
- Confirming adherence to country-specific guidelines established by Regulatory authorities such as the FDA, EMA, EU MDR, MHRA, TGA, HSA, PMDA, ANVISA, Health Canada, and others.
- Confirming adherence to country-specific guidelines relating to anti-kickback statutes and industry codes of ethics such as FTC, PhRMA, EFPIA, ABPI, PAAB, TGA, Medicines New Zealand, ANVISA, ANAMED, IFPMA, AIFA, AEMPS, Mdeon, and others.
- Validate proper use of claims and references. Claims are substantiated by relevant, appropriate references and scientific evidence, and efficacy or safety is not exaggerated.
- Validate a Fair Balance in the presentation of product risks and benefits, including any potential side effects or consequences of use.
- Evaluating the language, imagery, and other elements in the presentation of information to ensure clarity, accuracy, and no misleading information for target audiences.
- Verifying alignment of design and background elements with the content to avoid discrepancies (e.g., ensuring images of paediatric populations are not used in contexts where the product is not prescribed for that population).
- Accuracy of brand and company trademarks, service marks, device conformity certifications such as CE mark, UKCA mark, CCC, and others.
- Serve as subject matter experts (SMEs) on Regulatory matters during PRC meetings.
- Guide HA Submissions, such as OPDP/APLP 2253 or HA vetting of promotional materials.
- Advertisement and Promotional labelling Submissions to Health Authorities, such as OPDP/APLB Form 2253 to the US FDA in manual and eCTD formats.
Our team is highly innovative and can develop content for any platform or channel and hence our offerings/expertise are not limited to the above-mentioned, we can do a lot more!
- Review the drug-related content, including indications, dosage, instructions for use, and mechanism of action, to ensure appropriateness and accuracy as per the Approved Product Labeling, Prescribing Information (PI), SmPC, Patient Information Leaflets (PILs), Instructions for Use (IFU) and supported by proper clinical and scientific evidence.
- Evaluate the promotional material's alignment with current treatment guidelines and standards of care within the specific therapeutic area
- Customizing the review checkpoints to fit the therapeutic areas with guidelines or regulations for promotional activities, such as oncology or rare diseases
- Ensuring a "fair balance" between the presentation of risks and benefits in the promotional material, accurately reflecting potential drawbacks alongside advantages
- Clinical trials and other accurate, credible scientific data adequately substantiate claims.
- Verify that valid and reliable data support comparative claims with other products.
- Ensure that medical terminology and synonyms are thoughtfully selected for clear communication.
- Ensure target audience appropriateness (e.g., healthcare professionals, patients, caregivers) and that the materials are tailored to their understanding and needs.
- Collaborate with cross-functional teams on the Global to Local country-specific MLR review process and the Promotional Review Committee (PRC) process's current scenario assessment, perform gap analysis, and identify areas for optimization and harmonization.
- Setting up process SOPs, Work Instructions for MLR reviews, and KPIs to measure process efficiency.
- Experience defining MLR/PRC process workflows for LCM systems like Veeva PromoMats and MedComms.
- Facilitate the MLR review and PRC process for advertisements and promotional/non-promotional materials in LCM systems like Veeva PromoMats, Veeva MedComms, Pepperflow, etc.
- Create and distribute meeting agendas to stakeholders, participate in PRC meetings, and document minutes and decisions made by the PRC.
- Route promotional and advertisement material to reviewers in a prioritized and timely manner.
- Schedule and administer all concept, pre-submission, and live review meetings with stakeholders.
- Track material review approvals, ensure revisions are accurately incorporated.
- Ensures advertisement and promotional material meet submission-ready criteria.
Promotional Materials MLR Review
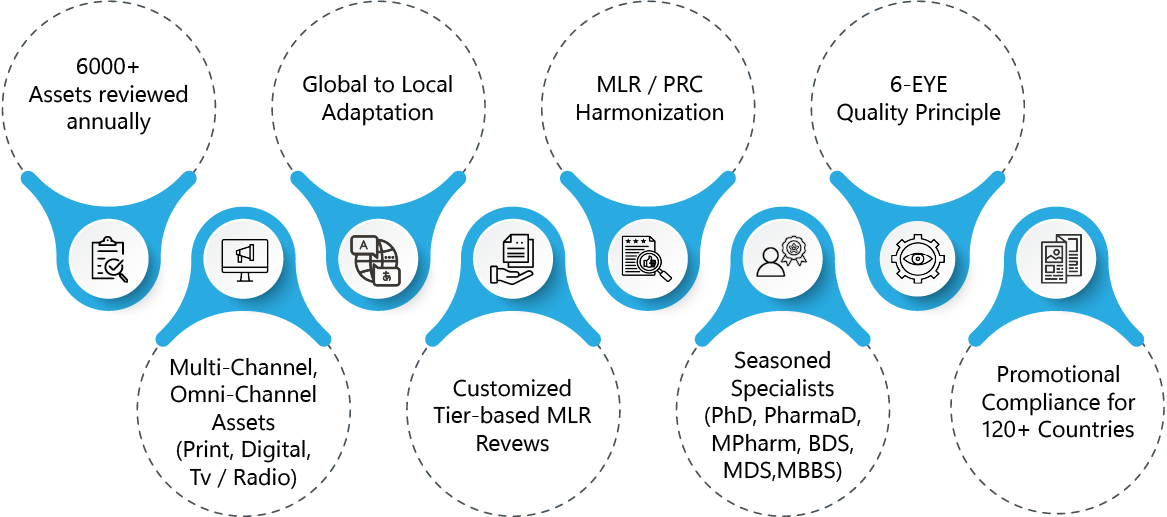
- 6000+ Assets reviewed annually for MLR Compliance
- Experience in Global to Local Promotional assets adaptation and MLR/PRC process harmonization
- 6-Eye Quality Principle for enhanced check on Regulatory compliance, adapted for therapeutic area complexities
- MLR Review Expertise Spanning a wide array of advertisements, promotional and non promotional assets for multiple communication channels – Digital, Print, Tv/Radio
- Customized Asset Complexity-based Tiered MLR review
- Promotional Compliance and MLR Review specialists with Industry experience and Clinical / Biomedical backgrounds (PharmaD, PhD, Mpharm, BDS, MDS, MBBS)
- Strategic local region-specific consultation on promotional material compliance for 120+ countries.
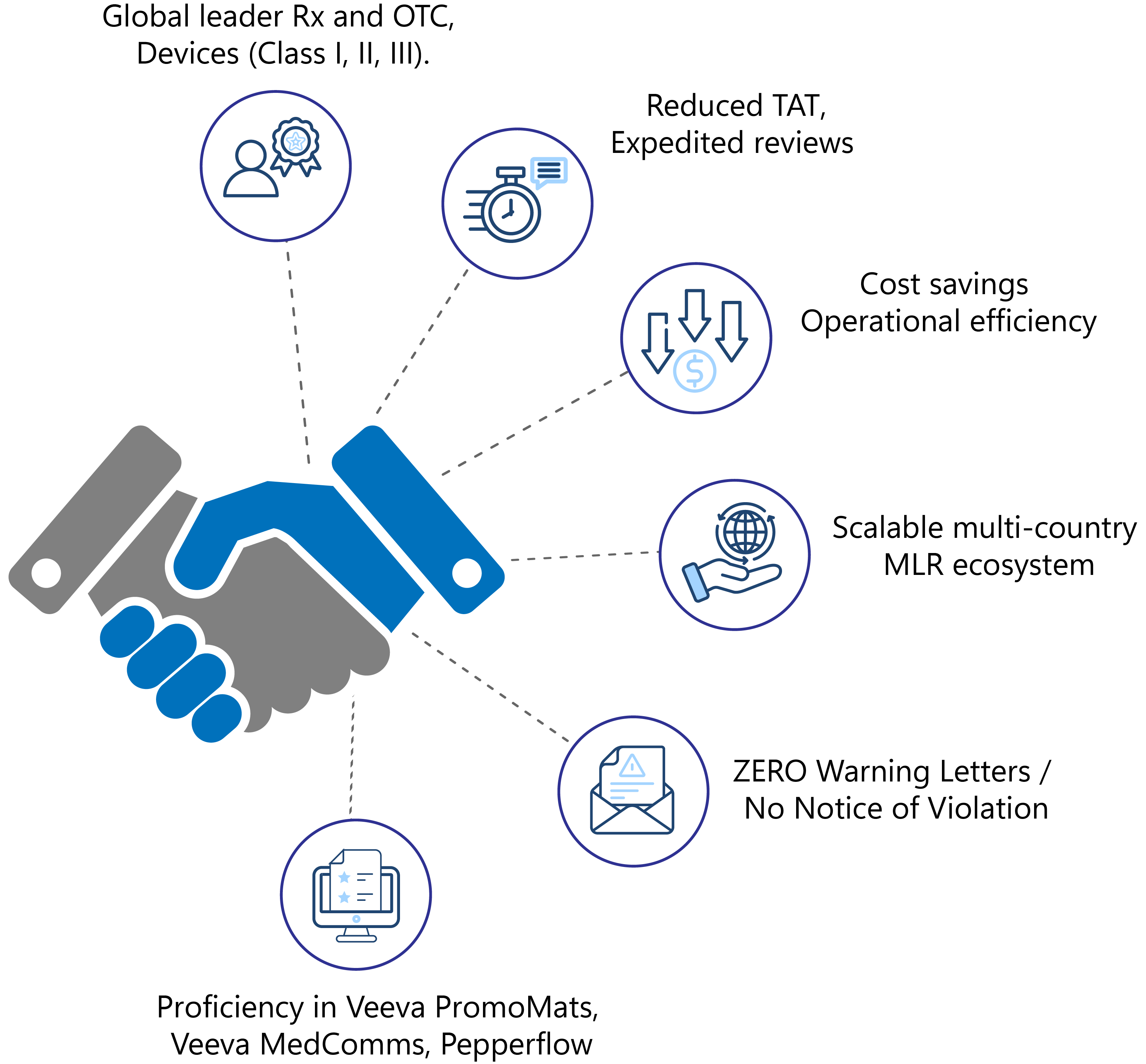
- Global leader in pharmaceutical, biopharma (Rx and OTC), and medical devices (Class I, II, III).
- Reduce overall cycle time for review
- Reduced TAT with expedited reviews to meet deadlines
- Significant cost savings and improved operational efficiency
- Scalable multi-country MLR ecosystem
- ZERO Warning Letters/No Notice of Violation
- Proficiency with MLR LCM systems like Veeva PromoMats, Veeva MedComms, Pepperflow, etc.