The biggest challenge that creative medical device Research and Development (R&D) programs face is that of balancing clinical evidence with early patient access. To address this, a new review system was proposed in Japan, to pace up device clearance while maintaining safety and efficacy. This benefits patients who are inflicted with life-threatening diseases that lack effective treatments.
- September 28, 2023 Medical Devices, Regulatory Affairs
- September 28, 2023 Medical Devices, Regulatory Affairs
As technology continues to advance, so do the medical devices that are used to sustain life. While in South Korea, various types of medical devices capable of communication have been developed, the development is accompanied by the risk of cybersecurity threats such as hacking of medical devices and information leaks.
- September 28, 2023 Medical Devices, Regulatory Affairs
Software as a Medical Device (SaMD) is an emerging category of medical devices that has been experiencing rapid growth in recent years. This category of devices includes medical device software applications designed for medical purposes, such as diagnosis, disease prevention, treatment, and monitoring.
- September 27, 2023 Cosmetics Regulatory Services, Regulatory Affairs, Regulatory Labeling
The cosmetic industry of Hong Kong is thriving, and it offers a diverse array of beauty and personal care products. Regulations and standards are in place to ensure the safety and efficacy of these products. Manufacturers and importers must understand the cosmetic Regulatory scenario in Hong Kong to ensure consumer protection.
- September 21, 2023 Regulatory Software & Services
AI is the new buzzword, and it does not seem like its buzz is getting any fainter any time soon. We all know that Artificial Intelligence (AI) and Machine Learning (ML) have been a transformative force across various industries, and the medical sector is no exception. In recent years, AI/ML has made significant strides in revolutionizing Regulatory operations within the medical industry.
- September 20, 2023 Pharmaceuticals, Regulatory Labeling
The development and approval of medications for children is a critical aspect of pediatric healthcare. However, due to ethical and practical considerations, clinical trials involving children are often limited. As a result, pediatric drug labeling plays a crucial role in ensuring the safe and appropriate use of medications in this vulnerable population.
- September 20, 2023 Regulatory Artwork Services, Regulatory Labeling
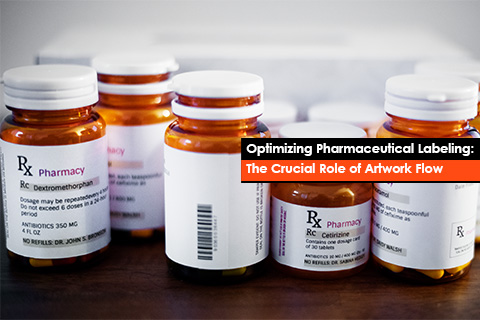
Quality is paramount in the pharmaceutical industry. From research and development to manufacturing and distribution, every step must adhere to the highest standards of accuracy to ensure patient safety and Regulatory compliance. One of the major aspects of this intricate process is pharmaceutical labeling, specifically the artwork flow.
- September 20, 2023 Pharmaceuticals, Regulatory Artwork Services
In the highly regulated pharmaceutical industry, the production of medicines and related products requires meticulous attention to detail and compliance with various regulatory guidelines. One critical aspect of pharmaceutical production is creating, approving, and managing artwork, including labels, packaging, and promotional materials.
- September 20, 2023 Publishing & Submissions
The ever-evolving Regulatory landscape demands businesses to adapt swiftly to new submission formats. Converting legacy data to meet these requirements in real time presents a complex and multifaceted challenge. It is in the best interest of the organizations to be able to convert their legacy data quickly and efficiently to new formats to comply. This can be a complex and challenging task, especially in real-time.
- September 20, 2023 Regulatory Affairs, Publishing & Submissions
The Clinical Trials Information System (CTIS) is a centralized database that supports the conduct of clinical trials in the European Union (EU). It was launched in January 2022 and is a key component of the Clinical Trials Regulation (CTR), which aims to improve the efficiency and transparency of clinical trials in the EU.