Food and Food Supplement Regulatory consulting
Freyr delivers comprehensive, end-to-end Food Regulatory consulting services tailored to the food and dietary supplements industry, enabling seamless global market access. From Food product classification services, ingredient and formulation compliance, and raw material specifications to claims substantiation, labeling, and novel food approvals, we ensure your product meets every Regulatory checkpoint. Our expertise extends to manufacturing facility registration, dossier compilation, and strategic health authority interactions. In addition, Freyr supports your sustainability goals through EPR, EUDR, and ecolabelling compliance. With unmatched Regulatory intelligence and global presence, we help you bring safe, compliant, and innovative products for Global Market Entry for Food Products.
Industry News
Food Supplement Regulatory Expertise
Navigating Regulations, Ensuring Innovation: Your Trusted Food Supplement Regulatory Experts
Food & Food Supplements Product Services
Categories Supported
Sustainability Services
Global Regulatory Intelligence
Make Regulatory Intelligence your secret ingredient to effortlessly navigate complex regulations on ingredients, claims, and more, ensuring global compliance for food and dietary supplements.
Country-specific Food Supplement Regulatory Consulting
Operating across continents, Freyr Solutions has established a strong global presence. With regional expertise and a deep understanding of local regulations, we deliver tailored solutions in key markets worldwide.
Africa & Middle East
Contact Us for Food Supplement Regulatory Expert Guidance and Support
Freyr's Approach to Food Regulatory
At Freyr, we understand the intricacies of global food and dietary supplement regulations, and we are here to guide you through the process of achieving Regulatory compliance for a successful market entry. Addressing the challenge of product classification, we navigate diverse jurisdictions with varying criteria, particularly in the complex realm of food supplement classification and ingredient assessment. Consumer safety, the paramount concern, involves adhering to stringent regulations to ensure the safety of food and dietary supplements. Moreover, evolving regulations add another layer of ongoing complexity to the process.
In regions that have distinct Regulatory requirements for food/dietary supplement products, developing a robust marketing strategy becomes essential for handling procedural challenges imposed by local regimes. The intricate nature of food and dietary supplement classification further underscores the importance of accurate interpretation of regulations to avoid critical mistakes. Freyr excels in providing global Regulatory Intelligence (RI) on food and dietary supplements, offering you a Regulatory roadmap for compliance, and successfully guiding you through your expansion plans in burgeoning health food markets. Our specialization encompasses food product registration, Good Manufacturing Practice (GMP) compliance, licensing, approvals, notification, Legal Representation (LR) services, Market Authorization Holder (MAH) services, license holding, imports and distribution, food product marketing strategy, Regulatory pathways, and comprehensive Regulatory strategies.

In the food industry, ensuring Regulatory compliance for products is vital. A key step is meticulously compiling a food/dietary supplement product dossier, gathering essential documentation for food and dietary supplement registration worldwide. This comprehensive dossier covers food registration, ingredient data, labeling requirements, and safety assessments.
Countries vary in terms of their respective food and dietary supplement classification, compliance, and registration requirements. Some countries mandate registration, while others focus on compliance without requiring formal registration. In countries with a registration-based system, products are categorized according to their risk levels, each requiring a distinct registration process. Additionally, terminology varies from one country to another, with some countries employing simple notification or approval language and others using more complex terms.
A thorough product dossier includes administrative, manufacturing, quality, control, and clinical data. Freyr aids in understanding and compiling the technical documents required for product registration in compliance with Health Authority (HA) regulations.
We help you streamline the process, assisting in gap analysis, document review, and dossier compilation for successful Regulatory submissions.

In the domain of food and dietary supplement registration and notification, numerous countries prefer local support to facilitate effective communication on product status and consumer safety. To address this requirement, companies often seek a Legal Representative (LR)/Market Authorization Holder (MAH)/Responsible Person (RP) post the registration stage for overseeing the product license, handling queries, and managing post-market obligations. In fact, the representative acts as a vital intermediary between manufacturers and the respective Health Authorities (HAs).
Freyr, with regional offices worldwide and an extensive partner network in 120 countries, provides LR services for food and dietary supplement registration. Operating in Europe, North America, Middle East and North Africa (MENA), Asia-Pacific (APAC), and beyond, we ensure seamless communication between our customers and Health Authorities (HAs). Our services guarantee compliance with country-specific regulations, oversee product registration, and maintain product data validity throughout the specified period.

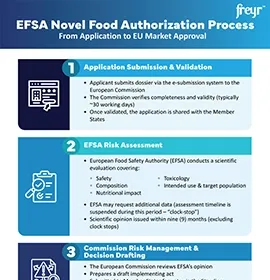
Novel food/ingredient registration involves submitting an application detailing the composition, production process, and intended use of the new food/ingredient to the specific Heath Authority (HA). This is followed by a rigorous safety assessment, evaluation of novelty, risk analysis, and decision-making regarding authorization. Post-market monitoring ensures ongoing compliance and product safety. The process aims to safeguard public health by ensuring that novel foods/ingredients meet the required safety standards before they are placed on the market.
Freyr assists in the registration process for novel food/ingredients, starting from conducting the preliminary feasibility assessment to determining whether the food/ingredient qualifies for novel food status, followed by a thorough review of its composition, production methods, and safety data. Freyr provides further support in conducting document reviews according to the guidelines of the respective Health Authority (HA), thus ensuring compliance. Upon completion of this entire process, Freyr submits the application for novel food registration on behalf of the customer and maintains ongoing communication and regular follow-ups with the respective HA to address any inquiries or requests for additional information, thereby facilitating a smooth registration process.