
Regulations stand crucial for any industry that functions in the Life Sciences domain, which is the third most heavily regulated sector after the Aviation & Financial sectors. Non-compliance with regulations comes at a high cost – the patients’ lives, heavy economic penalties, or loss of reputation. But challenges to this sector comes from various areas.
On one hand, the regulations are ever-evolving, and the authorities publishing them are many. On the other side, access to this critical information is often locked behind paywalls. Navigating this complex landscape requires proactive seeking out of data and continuously updating the knowledge base, which is best left to automation.
The need of the hour is a Regulatory Intelligence tool that compliments human wisdom with efficient robotic processes and artificial intelligence - ensuring organizations make informed and holistic decisions that are best for them and the people they serve.
Industry Challenges
- Lack of access to relevant information
- Constantly growing data volume
- Discrete sources of information
- Language barriers in non-English speaking countries
- Changing regulations with time
- Inefficient data curation processes – which are often manual
- No single platform to gather information
- Long turnaround times
- Unstructured data pool
Industry Needs
- Real-time extraction and curation of Regulatory requirements that are ready for consumption
- Track Health Authorities’ policies and guidelines
- Assess the impact of a change to a policy on the existing activities of the organization
- Provide market intelligence that is proactive and future-forward
- Auto-categorization of Regulatory information based on relevance and domain needs
- Replace inefficient manual processes with bots and other intelligent scraping tools
- Incorporate a recommendation system to drive decisions
- Automate summary and translations
Thus, the industry is ripe for AI (artificial intelligence) boom – where human intelligence will complement data-driven decision-making. The initiative represents the perfect (yet unexplored) use case for ML, Statistics, Text Analytics and NLP (Natural Language Processing). Hence, not only will ARI ensure that you are keeping up with the game, but the technology-centric innovation will also take organizations way ahead of the competitor curve.
Some of the use cases for having an advanced RI Platform in the Life Science industry are the following:
- Hastening submission processes
- Promptly responding to Health Authorities' deficiency mails & the evaluation process
- Holistic decision-making for new market entry or new product introduction
- Ensuring Regulatory compliance over the entire life cycle of the product
- Reducing inefficiencies and costs by cutting out manual processes
- Planning marketing activities to stay ahead of competitors
- Maintaining compliance in countries where information is scarce but the penalty for non-compliance is heavy
A truly advanced RI platform can reduce costs and create value.
Illustrating How Automation Can Improve Business Operations
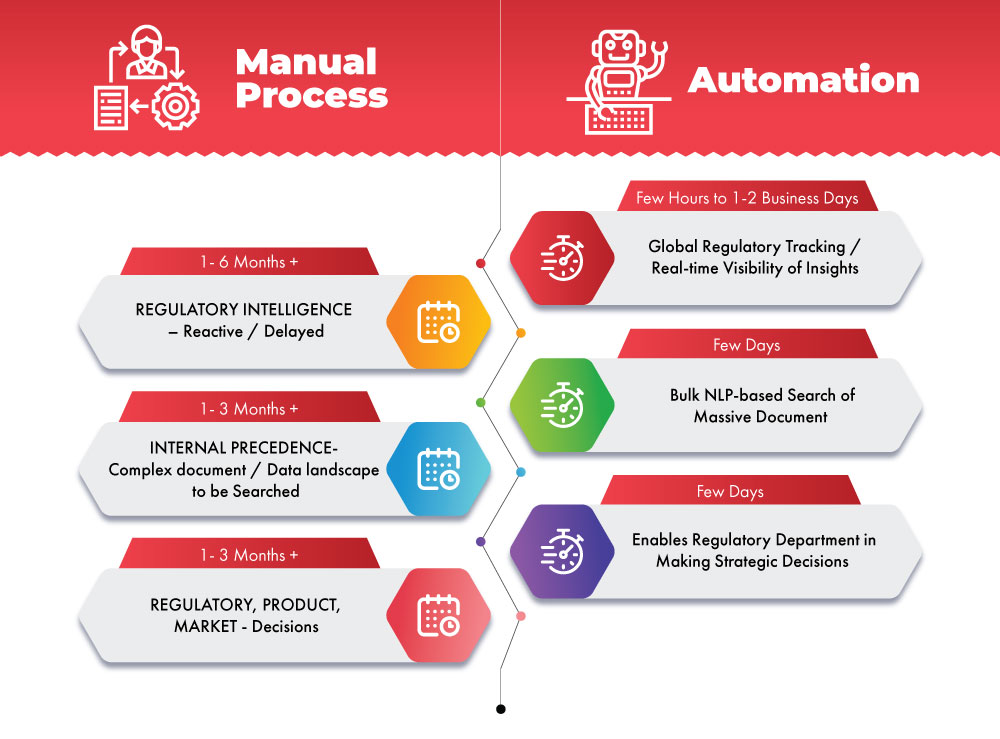
The AI/ML-enabled Next Generation Regulatory Intelligence System proactively streamlines critical decision-making processes and provides strategic advantages. Combining the power of data with advanced technology accompanies the life Science industry's needs.
Experience how Freyr IMPACT can be your intelligent compliance partner. Contact our experts here: Digital Life Science Regulatory Solutions & Software (freyrdigital.com)









