
The process of submitting Regulatory documentation to Health Authorities can be complex and time-consuming. Many organizations partner with a vendor that offers a submission tool to ease this process. With so many options in the market, it can be difficult to know who the right strategic partner is for your submission requirements.
This blog will discuss what you should consider when selecting an appropriate electronic submission service vendor. By considering the following factors, you can ensure that you find a vendor who can provide the right support, best procedures, and efficient tools for successful submission.
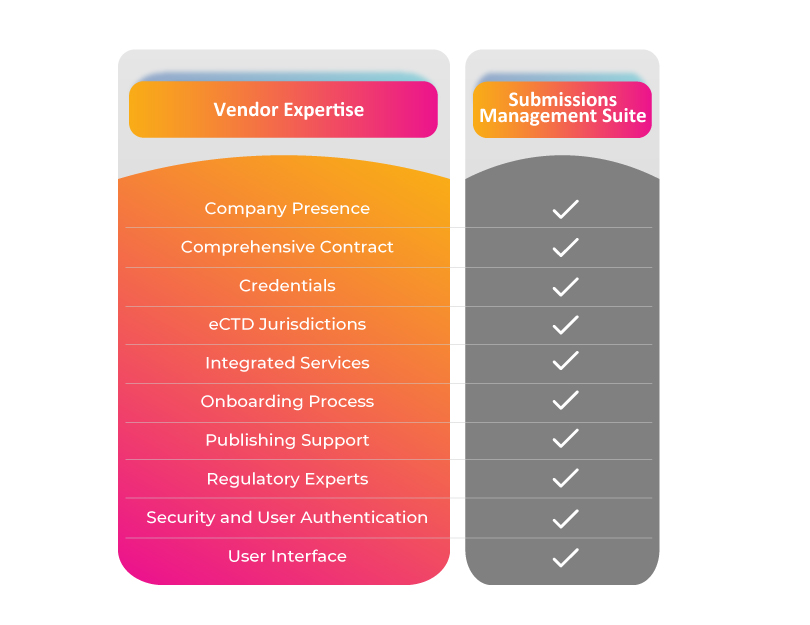
Company Presence:
Firstly, does the company have a strong presence in the submissions publishing space?
A well-established company with a track record of success stories is the foremost attention-seeking factor any organization would intend to look at because it holds not only market leadership but also broad expertise in the Regulatory operations and submissions space.
Comprehensive Contract:
Is the contractual agreement vendor friendly?
A clear agreement between the organization and the submissions vendor helps to ensure that both parties have understood the project scope. The contract should be clear and elaborate on roles, responsibilities, and timelines.
Credentials:
How essential is it to have strong credentials?
The major assets that can help to derive the selection of a vendor are case studies, success stories, testimonials, and clients. One can contact references to verify their quality of work. Thus, reliable credentials help decide on a vendor for the process of submissions.
eCTD Jurisdictions:
Are all the templates available to support various eCTD jurisdictions?
It is a huge plus! When the submission vendor has multiple templates to choose from. Such templates can be re-used to reduce the challenges faced in the submission components when each time a submission must be filed.
Integrated Services:
Will one vendor be able to offer all the support services?
A team full of submission experts guiding throughout the submission process and can support publishing activities through technical expertise is all an organization needs. If vital factors like teamwork, financial outlook, and partnering have been taken care of by using just one vendor what more can an organization ask for?
Onboarding Process:
How speedy is the onboarding process?
When it’s time to begin the process, selecting a vendor who can ramp up the onboarding activities smoothly could make a big difference between the missing and the meeting of a Regulatory submission deadline.
Publishing Support:
How flexible and scalable is the publishing support going to be?
Round-the-clock publishing support is every organization’s need to meet submission deadlines. An efficient vendor can help seamlessly move the project forward. Look for vendors that can handle several submission challenges with flexible software and sound support services. The vendor should be able to scale up and down as per the need of the organization to meet the most challenging deadlines.
Regulatory Experts:
Are they specialized Regulatory and consulting experts?
Look for vendors that help strategize and advise on the submission type. The Regulatory operations team must have thorough expertise in the submission publishing process and should know how to utilize technology to assist it.
Security and User Authentication:
How advanced is the security, and how strong is the user authentication?
Security is the priority and demonstrating that the software applications undergone the certification process is of utmost importance. Secure software is to be designed to secure the entire project data, including Advanced Encryption Standard (AES-256) encryption for data at rest and transfer.
User Interface:
Does the platform have an easy-to-use interface and innovative technology?
Faster and efficient utilization of technology to gain a quality output is the ultimate focus of any organization from their submission’s vendor. While employing a simple user interface, key aspects that any life science industry considers looking at include whether it would save time or improve accuracy and quality. The vendor should be able to provide the Regulatory operations team with efficient Regulatory submissions software.
In conclusion, when selecting an eCTD submissions vendor, it is important to consider factors such as efficiency, cost-effectiveness, and the ability to meet Regulatory submission deadlines. Freyr Digital meets all requirements and acts as a strategic partner in your Regulatory submissions journey. With an integrated product suite, including Freyr SUBMIT PRO, Freyr SUBMIT Track, and Freyr rDMS, you can efficiently handle all aspects of Regulatory data and document management. Book a demo with our in-house specialist to experience Freyr Digital’s benefits.









