
In Hong Kong, the approval process for rare disease orphan drugs, particularly those containing New Chemical or Biological Entities (NCEs) for life-threatening or severely debilitating diseases, is managed by the Pharmacy and Poisons Board of Hong Kong. Below is a step-by-step guide to navigating the process:
Step-by-Step Guide to Rare Disease Orphan Drug Approval
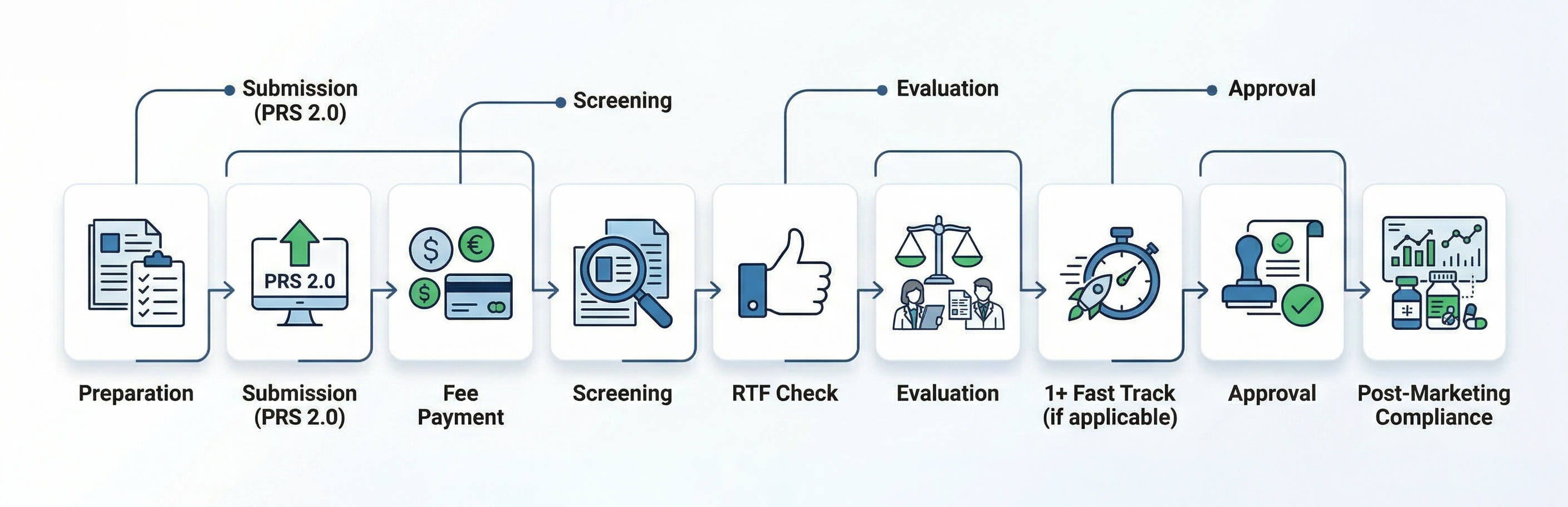
Preparation and Submission
Prepare the application including key documents:
- Expert evaluation reports and CTD documents on safety, efficacy, and quality
- Risk management plans (RMPs)
- Product labels and information leaflets
- Elemental impurity risk assessment reports
- Worldwide registration record
Submit your application for registration via the Pharmaceuticals Registration System 2.0 (PRS 2.0) on the Drug Office website. This is a critical first step for those registering orphan drugs for rare diseases in Hong Kong.
Payment of Fees
Pay the application fee through:
- PRS 2.0 (online) – via credit card or PPS
- In-person – using cash or cheque at the Drug Office
- Screening and Evaluation
- The Drug Office screens all submitted documents before accepting the application for evaluation.
- If documents are incomplete, a letter of deficiency will be issued.
- Refuse-to-File (RTF) Mechanism
- Effective 1 July 2024, the RTF mechanism ensures only complete and properly documented applications proceed to evaluation. A 60-day response period is provided for applicants to address any deficiencies identified during the initial screening.
- Incomplete applications will be refused at the screening stage. Failure to respond within 60 days will result in an automatic refusal of the application.
Enhanced Procedures for NCE Products
For NCE products under the “1+ mechanism” (for life-threatening or severely debilitating diseases), the Board has endorsed enhancements to facilitate and expedite the registration process—particularly beneficial for orphan disease drugs.
- Post-Approval Requirements
- After product registration:
- Comply with pharmacovigilance requirements
- Report serious adverse drug reactions (ADRs)
- Implement and maintain risk management plans
- After product registration:
Appeals
If aggrieved by a committee decision, the applicant may appeal to the Pharmacy and Poisons Appeal Tribunal.
Timelines
The Department of Health aims to complete the registration process around 9 months after submission, provided all documents are complete. This helps bring drugs for rare diseases to market efficiently.
Consultation and Feedback
The Board may conduct consultations with stakeholders and industry experts during the evaluation and policy implementation stages.
Continuous Updates
Stay informed by Regularly checking:
- The Board’s official website
- Announcements from the Drug Office
Flowchart
How Freyr Can Help
Freyr offers end-to-end support for registering orphan drugs in Hong Kong, including dossier preparation, PRS 2.0 navigation, and post-approval compliance. With local expertise and a strategic approach, we help ensure faster, compliant market entry for orphan drugs for rare diseases.










